Acidity is a vital part of food safety and taste. A sharp twist of lime or a splash of vinegar can help to turn a palatable dish into an incredible one. In food manufacture, control over acidity is absolutely essential in ensuring a product that’s consistent in flavour and safe to eat. This is done through careful monitoring of pH levels, via handheld pH meters.
What is pH?
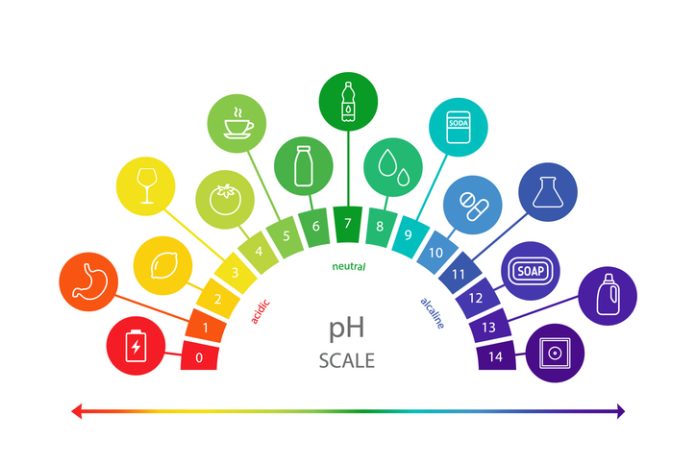
The acidity or alkalinity of a solution is measured on a scale from 0 to 14. In the middle is a pH of 7, which is considered neutral. While there are other systems for measuring acidity, pH is the most common. Much like the decibel scale used to measure sound volume, pH is logarithmic, which means that small changes in value can lead to big ones in the real world. A change of 0.3 ph means double (or half) acid concentration. So, if a substance goes from pH 7 to pH 6.1, it’ll become eight times as acidic.
pH in Dairy
Several good examples of the importance of pH come from the dairy industry. Most milk has a pH of around 6.8, making it very slightly acidic. When the pH of milk is different than this, it might indicate a problem with the cattle that produced it: certain sorts of infection will make milk more acidic.
The acidity of milk will also determine the quality of the cheese that it goes on to make. While the cream is being matured and milk is being soured, there’s a chance for harmful bacteria to multiply. This can be controlled by keeping the pH value closer to 5. A similar approach is taken during the manufacture of butter: a higher pH, closer to that of drinkable milk, will yield a sweeter butter; a lower pH will generate sour butter. The pH is typically influenced through the addition of citric acid extracts (which is why so many dairy products include acidic additives).
Some dairy products, like yogurt, must be made especially acidic. But if fruit is going to be added to the yogurt in the pot, then it will need to be of the same acidity in order to avoid an adverse reaction.
pH in Meat and Fish
pH is often used to assess the quality of meat when it is first brought into a factory. The figure will help to give an idea of the quality of storage further up the supply chain. Different cuts of meat contain muscular tissue and fat in different proportions. When the pH is too high, the quality of the meat will deteriorate – it won’t become as dark, and some aroma will be lost.
Certain sorts of meat and fish, like fillet beefsteak, oysters, and salmon, are often consumed raw. This makes screening for toxins especially important. In the case of shellfish, this means several rounds of high-pressure washing. Changes in the acidity of the water indicate whether pathogens have been successfully removed.